This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (CC BY).
ORIGINAL RESEARCH
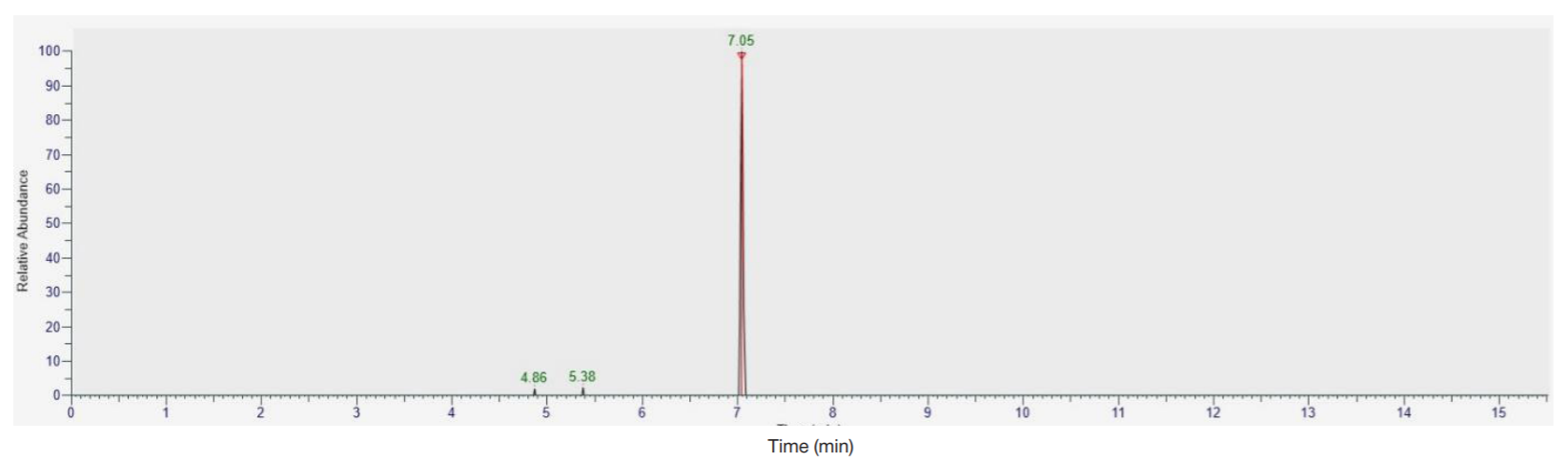
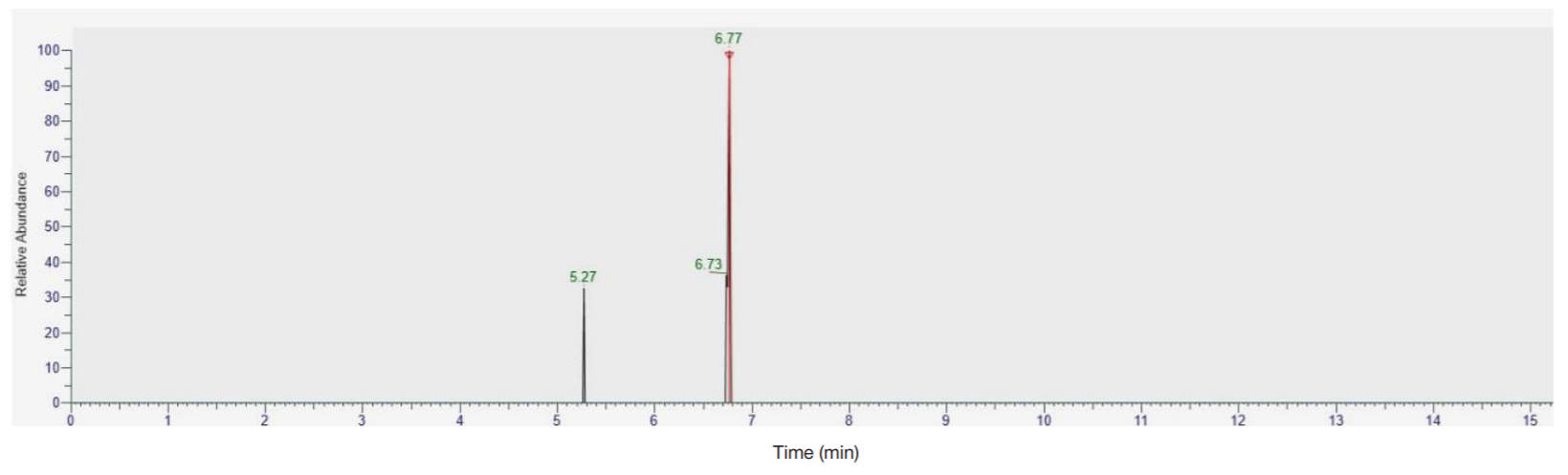
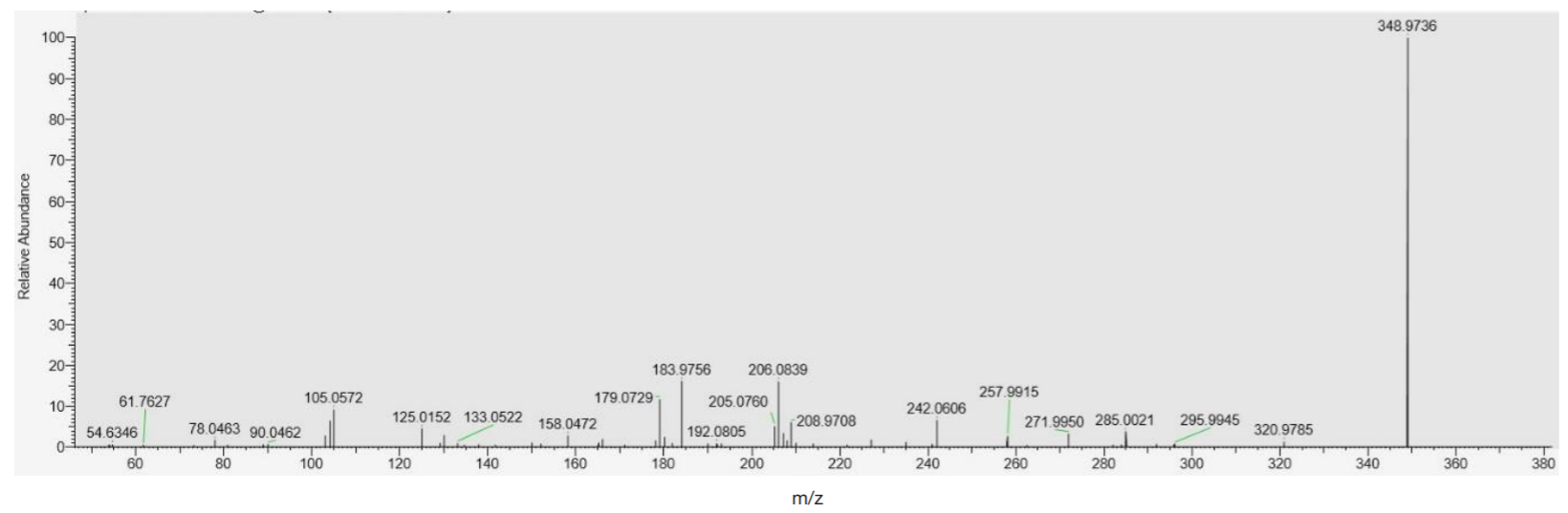
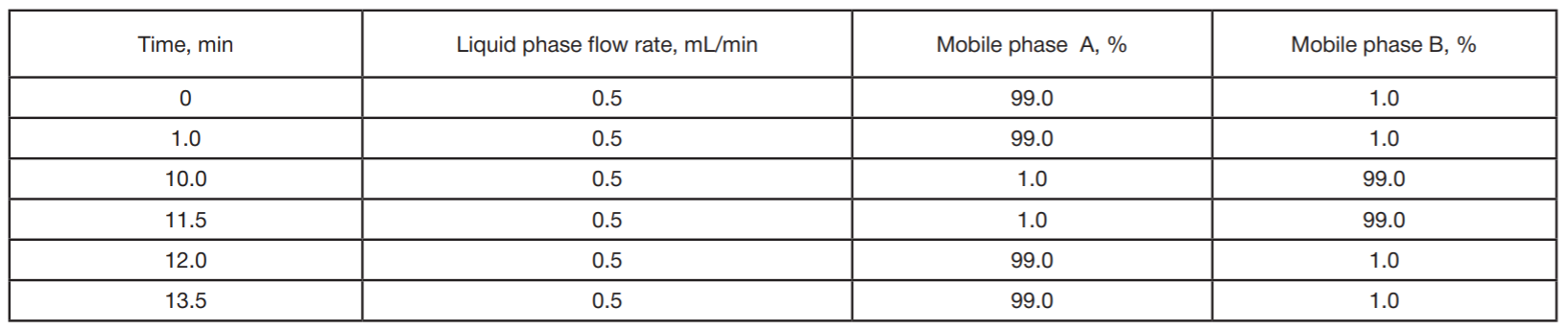
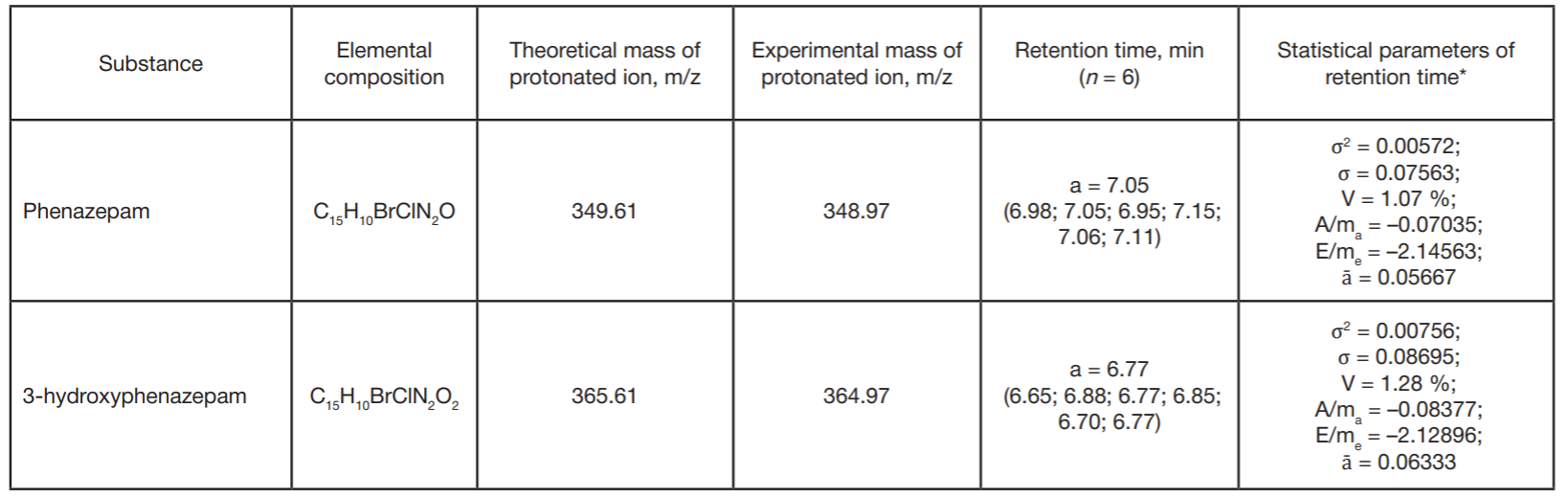
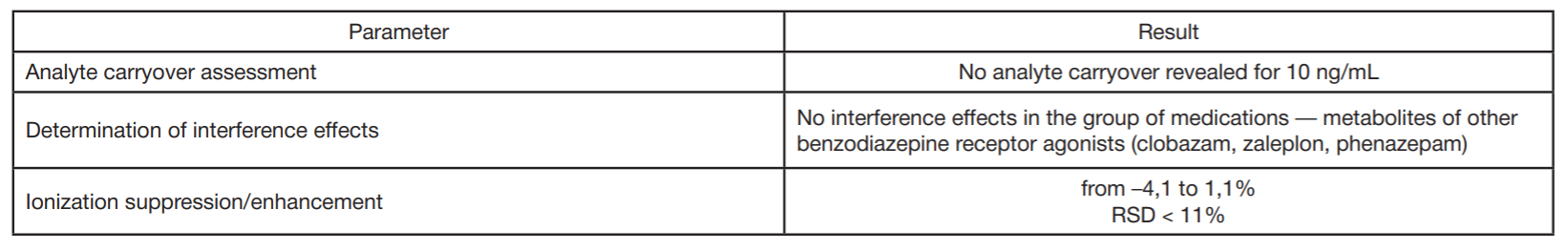
Detection of ultra-low concentrations of bromodihydrochlorophenylbenzodiazepine (phenazepam) and its metabolites in biological objects
1 Russian Center of Forensic Medical Expertise, Moscow, Russia
2 Peoples’ Friendship University of Russia, Moscow, Russia
3 Sechenov First Moscow State Medical University, Moscow, Russia
4 Bureau of Forensic Medical Examination, Moscow, Russia
Correspondence should be addressed: Roman A. Kalekin
Polikarpova, 12/13, Moscow, 125284, Russia; ur.ems-cr@ajimih
Author contribution: Volkova AA, Kalekin RA, Orlova AM, Astashkina OG — data acquisition, manuscript writing; Volkova AA, Kalekin RA, Moskaleva NE, Markin PA — experimental procedure.
Compliance with ethical standards: the study was planned and conducted in accordance with the requirements of the Declaration of Helsinki of the World Medical Association (2000) and subsequent revisions thereto; the informed consent was submitted by all study participants.